 Image 1 of 3
Image 1 of 3

 Image 2 of 3
Image 2 of 3

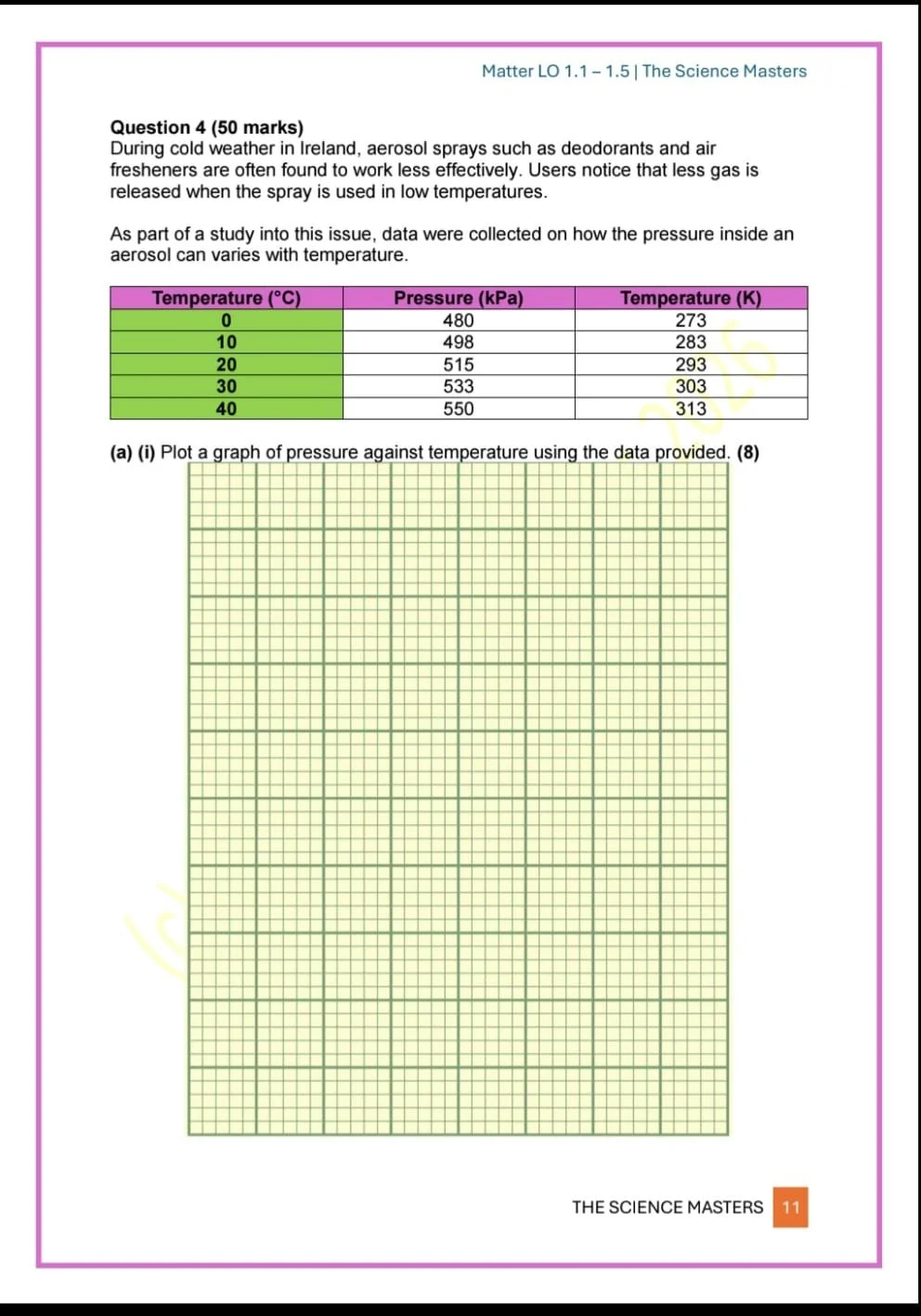 Image 3 of 3
Image 3 of 3




Leaving Certificate Chemistry Higher Level Strand 1 Complete Unit - 230 Page Exam Pack - Matter, Atomic Structure, Stoichiometry & Periodic Trends
Leaving Certificate Chemistry Higher Level – Strand 1 Complete Unit Pack
Thank you for reading this post, don't forget to subscribe!
This comprehensive 230-page resource provides full coverage of Strand 1 of the new Leaving Certificate Chemistry specification. It is designed to support both teaching and revision through structured, exam-style questions and detailed marking schemes.
Each learning outcome is developed through realistic contexts, scaffolded questions and repeated exposure to key concepts, helping students build confidence and mastery across the full strand.
What’s Included
230 pages of exam-style questions and structured answers
Full marking schemes for all questions
Coverage of all Strand 1 learning outcomes
Real-world contexts and applied chemistry scenarios
A mix of short-answer, structured and extended-response questions
Suitable for class tests, homework, revision or mock exams
Learning Outcome Coverage
LO 1.1 – Matter and the Kinetic Theory
Particle theory of solids, liquids and gases
Diffusion and Brownian motion
Effects of temperature on particle movement
Gas pressure, volume and temperature relationships
Real-life applications including aerosols, tyres and gas systems
Experimental design and evaluation
LO 1.2 – Atomic Structure and Spectroscopy
Structure of the atom and subatomic particles
Isotopes and relative atomic mass
Emission spectra and electron transitions
Relationship between energy, frequency and wavelength
Mass spectrometry and data interpretation
Development and limitations of atomic models
Quantitative Chemistry – The Mole and Stoichiometry
Mole calculations and Avogadro’s number
Formula units, atoms and particles
Balanced equations and mole ratios
Gas volume calculations (including STP)
Conservation of mass and chemical laws
Real-world applications (medicine, industry, food science)
Multi-step problem solving and exam-style calculations
LO 1.3 – Periodic Trends and Chemical Behaviour
Structure of the periodic table (groups, periods, blocks)
Ionisation energy trends and explanations
Atomic radius and shielding effects
Electronegativity and bonding
Reactivity of Group 1, Group 2 and Group 17 elements
Transition metals and noble gases
Displacement reactions and real-world applications
Why This Resource Is Effective
Reinforces key concepts through repeated exposure in different contexts
Develops both conceptual understanding and exam technique
Builds confidence with structured, scaffolded questions
Aligns with the new specification approach and style
Allows teachers to assess learning across an entire strand
Ideal For
Leaving Certificate Higher Level Chemistry students
Teachers delivering Strand 1 of the new course
Revision classes and exam preparation
Continuous assessment and class testing
Format
PDF download
Print-ready
Can be used as a full unit booklet or split into topic tests
Leaving Certificate Chemistry Higher Level – Strand 1 Complete Unit Pack
Thank you for reading this post, don't forget to subscribe!
This comprehensive 230-page resource provides full coverage of Strand 1 of the new Leaving Certificate Chemistry specification. It is designed to support both teaching and revision through structured, exam-style questions and detailed marking schemes.
Each learning outcome is developed through realistic contexts, scaffolded questions and repeated exposure to key concepts, helping students build confidence and mastery across the full strand.
What’s Included
230 pages of exam-style questions and structured answers
Full marking schemes for all questions
Coverage of all Strand 1 learning outcomes
Real-world contexts and applied chemistry scenarios
A mix of short-answer, structured and extended-response questions
Suitable for class tests, homework, revision or mock exams
Learning Outcome Coverage
LO 1.1 – Matter and the Kinetic Theory
Particle theory of solids, liquids and gases
Diffusion and Brownian motion
Effects of temperature on particle movement
Gas pressure, volume and temperature relationships
Real-life applications including aerosols, tyres and gas systems
Experimental design and evaluation
LO 1.2 – Atomic Structure and Spectroscopy
Structure of the atom and subatomic particles
Isotopes and relative atomic mass
Emission spectra and electron transitions
Relationship between energy, frequency and wavelength
Mass spectrometry and data interpretation
Development and limitations of atomic models
Quantitative Chemistry – The Mole and Stoichiometry
Mole calculations and Avogadro’s number
Formula units, atoms and particles
Balanced equations and mole ratios
Gas volume calculations (including STP)
Conservation of mass and chemical laws
Real-world applications (medicine, industry, food science)
Multi-step problem solving and exam-style calculations
LO 1.3 – Periodic Trends and Chemical Behaviour
Structure of the periodic table (groups, periods, blocks)
Ionisation energy trends and explanations
Atomic radius and shielding effects
Electronegativity and bonding
Reactivity of Group 1, Group 2 and Group 17 elements
Transition metals and noble gases
Displacement reactions and real-world applications
Why This Resource Is Effective
Reinforces key concepts through repeated exposure in different contexts
Develops both conceptual understanding and exam technique
Builds confidence with structured, scaffolded questions
Aligns with the new specification approach and style
Allows teachers to assess learning across an entire strand
Ideal For
Leaving Certificate Higher Level Chemistry students
Teachers delivering Strand 1 of the new course
Revision classes and exam preparation
Continuous assessment and class testing
Format
PDF download
Print-ready
Can be used as a full unit booklet or split into topic tests
